Clinical Trial Abundance, Made in China
Flying to China for cancer care
A guest post by Jacob Stern:
I work with Sid Sijbrandij, a technology entrepreneur who has taken a radically personalized and high-agency approach to fighting his osteosarcoma (bone cancer). Before meeting Sid, I was a product lead at 10x Genomics, a sequencing technology company developing novel tools for understanding biology. Sid was the first person I met who had used 10x tools to inform their care. I now run the enterprise of Sid’s care, pursuing a strategy of maximal diagnostics, making personalized therapeutics, and doing treatments in parallel rather than one at a time. Against the odds, Sid has had no evidence of disease for almost a year now. We are scaling this approach for others, both by starting companies and through philanthropic efforts.
Elliot Herschberg wrote an excellent and approachable post on Sid going “Founder Mode” on his cancer on his blog, Century of Biology. We recently gave a talk at the OpenAI forum on Sid’s journey and our approach. More details can be found at sytse.com/cancer, and 25TB of data and Sid’s treatment timeline are available open source at osteosarc.com.
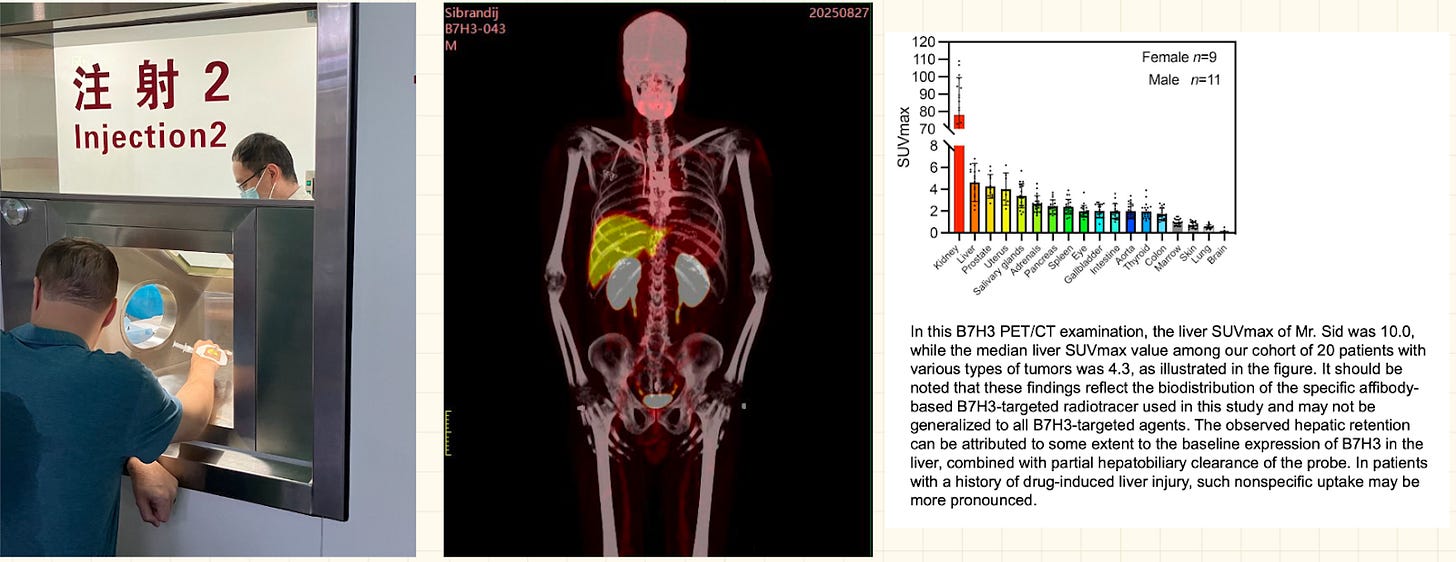
Last August, Sid Sijbrandij and I traveled to Beijing for an experimental scan to look at a biomarker that’s specifically upregulated in his cancer.1 At that time, the only place we could do this was in China, using a molecule developed by Yang Zhi (杨志)’s group at Beijing Cancer Hospital. So that’s where we went.
We were stunned. The whole experience — from international patient check-in, to preparation of the radiotracer, to injection, to imaging, to discussing the result with the physician, to leaving with a glossy printout of the whole-body scan — took two hours. Even in Germany, where clinics are experienced in using developmental tracers, this process would take most of a day. Beijing broadly and the hospital specifically were surprisingly straightforward to navigate for foreigners such as us who speak no Chinese.
This experience inspired me to return to China in search of a deeper understanding of what is happening at the forefront of biotech and medicine. I often read and hear that it is becoming more difficult for American biotech to compete with what’s happening in China. I wanted to understand specifically what was going on, and what the implications were for a patient seeking the world’s most innovative care.
I spent a week in China at the end of March, visiting 5 cities in 6 days.2 I had over 25 meetings with biotech companies, investigators, contract research organizations (CROs), and contract development and manufacturing organizations (CDMOs). I came away impressed. Medical tourism is likely to invert, with patients flying to China to seek cutting-edge care. And I hope that we in America can learn from the sensible steps the Chinese ecosystem has taken and speed up our own innovation cycle. Patients deserve it.
A Marketplace of Reputation
The “investigator-initiated trial” (IIT) is an important fundamental concept to understand. Through IITs, individual physicians at major hospitals in China can propose and run studies for cell and gene therapies under the oversight of local scientific and ethics committees. There’s no need to clear a single, centralized national gate before enrolling patients. Compare that to the United States, where early trials are usually company-driven and require formal approval from a national regulatory body (like an IND filing with the FDA) before anything can begin. The tradeoff is pretty straightforward: the US system emphasizes uniform standards and upfront rigor, while China’s IIT model pushes decision-making closer to the doctor and the patient, making it easier to start trials quickly and iterate as data comes in. Carvykti (ciltacabtagene autoleucel) is perhaps the most striking example of what this model can produce. The BCMA-targeting CAR-T therapy first entered human clinical trials through an IIT in China, and has since gone on to reshape the standard of care for relapsed or refractory multiple myeloma — a disease where treatment options had long been limited for patients who had already cycled through multiple prior lines of therapy.
For more on Carvykti and China’s biotech coming of age see this feature we ran last year:
Biotech
In 2011, China’s drug regulator cleared the nation’s first home-grown targeted cancer pill. Fourteen years later, a Chinese bispecific antibody is aiming to knock the world’s top-selling oncology drug off its perch.
Based on what I heard on the ground, it takes about 6 months to go from a first conversation between a doctor and a patient to that patient getting dosed, and people noted that exciting programs with support from senior investigators can go even faster. This means new therapies can get to patients faster, and companies and physicians start learning from and improving the underlying therapies earlier in their development.
One of the defining features of IITs is that reputation acts as the primary coordination mechanism, and that in turn helps enforce safety. Investigators are highly attuned to reputational risk. Because a death or a serious adverse event can have lasting professional consequences, they design protocols carefully, demand strong supporting data, and prioritize projects they believe are both safe and scientifically credible. At the same time, relationships play a central role. Since every trial involves uncertainty, investigators tend to work with collaborators they trust from prior experience. As a result, trial opportunities are allocated less by price and more by a combination of trust, track record, and perceived scientific promise. And the balance of supply and demand is such that academics with the platforms to do IITs and recruit patients quickly have many options, with both local and global biotechs approaching them with ideas, so they can be choosy.
The emergent system is a more pragmatic and cost-efficient one than what we see in the US for equivalent trials. In China, institutional ethics committees set their own manufacturing and pre-clinical data standards for project initiation. Because reputation is on the line, the standards are strict but sensible. They demand robust manufacturing controls and toxicity studies, but not at the level typically required in the US for trials of this stage. While the details can differ, the standards are similar enough — and the volumes at each institution are high enough — that CROs and CDMOs have set up IIT platform processes sufficiently mature that they could quote me approximate prices.
China’s State Council has recently adopted Decree 818 (国务院令第818号) to streamline IITs for cell and gene therapies. Prior to this regulation, IITs were popping up everywhere (particularly around regenerative cell therapies), leading to uneven data quality. With the goal of making data quality more systemically robust, 818 restricts the authority to run IITs to a pre-selected set of Tier 3 hospitals and requires Good Clinical Practice (GCP) certification for investigators. Interestingly, 818 opens the door to bring therapies to market very very quickly. Once ~10-15 patients have been treated with a therapy at a given hospital, that hospital can apply for the right to charge patients for access to that therapy. Essentially, the combination of therapy and institution is being approved. Data across institutions can also be leveraged for national approval down the line.
All of this makes sense! The system leans on the reputational sensitivity and naturally risk-averse incentive structure of academic medicine to regulate which medicines move forward to human trials. By putting trust in clinicians’ and hospitals’ judgment, the system is able to bring therapies to patients quickly.
Momentum is All That Matters
On my trip, I was repeatedly quoted a timeline of 18 months from a company having an idea for a therapy to testing it in a patient. My lived experience from my week on the ground backs up this speed. I experienced a sense of urgency at every level. Not just from start-up companies themselves, but also the ecosystem of third-party vendors that perform services for these companies.
During a visit with a CDMO focused on cell therapy manufacturing in Suzhou, I asked the business development rep giving the presentation about the company’s experience with non-viral gene editing. He picked up his phone. As we were preparing to leave 10 minutes later, the principal scientist responsible for the non-viral editing platform caught us by the door. He answered my questions, and we figured out the next steps to evaluate the suitability of their platform for the non-viral editing approach our collaborator is using.

In Shenzhen, I made a curiosity-driven comment about the instrumentation being used in an automated data foundry we visited. Two hours later, the head of the instrumentation company was waiting for us at the coconut chicken place we visited for dinner. If I wanted to put together a lab, he could do it.
A friend of a friend joined us in the afternoon in Shanghai. At that point, we’d only exchanged about two sentences in direct conversation, but she was there as I discussed supply chain considerations for personalized medicine projects with my main host. As I headed to the train station that evening, I got a WeChat message from an Executive Director at ATLATL, a well-connected incubator in Shanghai that serves as somewhat of a “scientific embassy” for international biotechs looking to explore doing business in China. The friend of a friend had suggested we meet. The next day, I was in the ATLATL offices for lunch with their founder, Dr. PC Zhu 朱鹏程.
These experiences are reflective of the connectedness and sense of urgency I saw at every step during my week in China. Whether with CROs and CDMOs in Suzhou or deep elements of the reagent and instrumentation supply chain in Shenzhen, local ecosystems were dense and highly connected. Competition is fierce at every level — destructively so, according to many people I talked to. Quality is very high for those in the know. People are responsive and flexible.
Labor costs, medical costs, and infrastructure costs are lower compared to the US and Europe. There’s apparently a local discount, too — I heard from one company with operations in both the US and China that the Chinese operation’s quotes from local providers are half what gets quoted to American companies (this gives them an advantage in capital efficiency). But the most striking dynamic I observed was the speed. For companies that know how to navigate (read: have relationships, know whom to trust, and possess pre-built trust with those people), there’s a vibrant, redundant, end-to-end supply chain that can be tapped on demand with a high degree of responsiveness.
The ability to go from zero to patient data in 18 months is an advantage that will compound, as companies and the ecosystem writ large will be able to get to the real learning (testing drugs in patients) faster and iterate. Many of the companies I talked to were primarily (if not solely) funded by local capital markets and domestic government support. But the local Chinese pharmaceutical market is not enormous, with prices substantially lower than in Western markets and many patients paying for drugs out of pocket. I got the sense that the ecosystem sees preclinical development and clinical data generation as an important export market, with China serving as the innovation and proof of concept generator for medicines that will help patients around the world. Capital is already starting to flow to support this vision. At multiple stops, my visit was preceded or followed by VC firms and multinationals looking to feed and tap this innovation engine. This is good! Humanity needs more medicines — and right now, the path of least resistance to generate more medicines seems to lead through China.
A Note on Good Intentions
In Shanghai, I met the founders of a company working on pH-sensitive modulators of oxidative phosphorylation as therapies for chemo- and radiation-resistant cancers, based on work the Chief Science Officer (who is also a professor at a local university) had done during his postdoc in the US. They’ve focused on high-grade gliomas, and we met because they think their approach may be a good fit for osteosarcoma as well. They plan to open their first clinical trial in early summer in Shanghai and Beijing.
I mentioned that I am in touch with a glioblastoma patient who’s going ‘founder mode’ on his own cancer, and asked if it was possible for an international patient to access their medicine. The two founders of the company looked at each other a bit sheepishly and then shared their story. A friend of one of the founders, also a biochemistry professor, reached out to them on behalf of his mother. She had high-grade glioma, was in a coma, and was running out of options. The founders were hesitant, but the professor insisted they try. So they did, outside the auspices of any official trial. The protocol was 5 days of temozolomide, followed by 10 days of the experimental therapy. The mother is on her third dose of the drug. She emerged from her coma after the first dose. The company doesn’t have trial-grade evidence of the efficacy of their drug, but they do have more confidence. The professor-friend is grateful.
I share this story because I found in China what I find everywhere — people do biomedical research and drug development because they want to help patients. Scientists were friendly, helpful, and precise. People readily made introductions. They offered to help navigate further. They want to help patients.
I would personally feel comfortable flying to China for care. If a loved one or I got cancer, I would look at the IITs happening in China (with local help to try to get the full menu of what’s available). There’s a reasonable chance the most innovative option is being developed there.
Closing Thoughts for America
One of the smartest, most innovative scientists I spoke with on my trip noted that the process China currently follows is inspired by the one America once used for cell and gene therapy. As Dr. Ruxandra Teslo has very cogently laid out in her work on Clinical Trial Abundance, the early work on CAR-T’s in labs like Carl June’s at the University of Pennsylvania followed a playbook that seems similar to what’s happening in China now. Meanwhile, efforts to extend the efforts that led to the Baby KJ gene editing triumph seem to be running into stringent standards that are unrealistic for academic groups or narrowly scoped efforts to overcome. Dr. Teslo has put forward a number of specific policy proposals that would help America return to agility in early-stage clinical trials. I suspect we could lean more on a marketplace of reputation to keep clinical research in check in the US. The Clinical Trial Notification Pathway recently proposed by the FDA would be a good step in this direction.3 Some states, including New Hampshire, Montana, and elsewhere, are also moving in this direction; if not precluded federally, state-level innovation could serve as a laboratory of governance to test different versions of reform prior to wider-scale implementation.
It was interesting to observe that the expanded access/single-patient IND pathway is more suited to flexibly get an individual access to a potentially important treatment than anything I heard about in China. It was encouraging to see Dr. Marty Makary say recently on X that he’s signed every compassionate use request that’s crossed his desk. This is great! We should continue leaning in on single-patient INDs, with situation-appropriate standards that reflect the risk of inaction for a patient in dire straits.
How can America go faster? The default is to leverage the CRO/CDMO infrastructure that’s available in China to develop Western IP, which is clearly happening. What about parallel infrastructure? My mind goes to companies like Plasmidsaurus, Adaptyv, and Aequita, which are building highly automated, fast, pay-by-credit-card offerings for specific high-volume assays, and earlier-stage analogs in manufacturing like Nature’s Toolbox, Harton, and Exthymic. I’d love to know what else is out there.
It is ironic to me that the ‘marketplace of reputation’ that seems to govern China’s IIT ecosystem is more market-oriented than the regulatory apparatus we use to govern early-stage trials in the US. Every system has its strengths and drawbacks, China’s included. The parts I saw up close show how the Chinese ecosystem is leaning into its strengths — velocity of science and engineering, urgency, close-knit relationships within the ecosystem, compassion for patients. I’m hopeful that, as a country, we can reflect on and actively lean into our strengths as an ecosystem too.
For more, please visit sytse.com/cancer. Please write to us with thoughts or questions at cancer@sytse.com.
While Sid has no evidence of disease, we want to use biomarker-targeted PET tracers for imaging — both to look for potential recurrence, and to do personalized biodistribution analysis of druggable targets. B7-H3 was a top target for us, as his cancer has high expression of B7-H3; were his cancer to return, we would think of treating it with a B7-H3 targeted agent, possibly with a highly potent CAR-T that Kole Roybal and his group at UCSF are developing.
Hong Kong, Guangzhou, Shenzhen, Shanghai, Suzhou
See page 26


Nice to see some appreciation of "Chinese characteristics".
The IIT model speed advantage is real, but the longer-term story is data accumulation. Running trials faster and earlier means generating patient data at a pace the US system structurally cannot match especially for rare diseases and early-phase experiments that would never clear FDA IND review at scale.
This feeds forward into medical AI in ways that are not fully appreciated yet. ByteDance Health, Baidu Health, and a dozen newer startups are training models on clinical datasets that dwarf what any Western health system has centralized access to. HIPAA-era fragmentation, institutional IRB silos, and restrictions on cross-hospital data sharing create training data deserts that Chinese developers simply do not face. DeepSeek medical reasoning being strongest on Chinese disease profiles is not a coincidence.
The inversion the author describes is the visible tip. The less visible story: every trial generates training data for the next generation of Chinese medical AI, compounding an advantage that the US regulatory framework actively prevents building.